REMAKING THE BRAIN
Georgina Ferry
Transplants of hearts,livers,kidneys and other organs have meant a new lease of life for thousands of people.But brain transplants still belong to the realms of sceince fiction.Or do they?
Hundreds of people have undergone operations during the 1800s to place cells in their brain which do not belong there. The aim is to reduce the symptoms of an incurable brain disease. Given the enormous complexity of the brain - billions of highly specialised cells, each with up to a thousand connections - does such an approach stand a chance of success?
A brain transplant, or more properly a brain graft, is a very different matter from other types of transplants. For a start the surgeon is not transplanting a whole organ. Replacing a kidney is effectively a plumbing job, rather like replacing a filter in a domestic water supply. Brain grafting is more like electrical engineering.
The brain consists of interconnected circuits that influence each other's activity. If one type of circuit is damaged, other, connected circuits may no longer function properly, even if they themselves are undamaged. The principle behind grafting is to replace the faulty circuits, so that the whole system can function once more.
Humans suffer from various diseases which appear to damage the brain selectively attacking a restricted area or population of cells. Among the most common are: Parkinson's disease, which affects muscle coordination, Alzheimer's disease, which causes memory failure in elderly people and Huntington's disease, which is hereditary and affects both movement and mental state in middle life. Strokes are not selective in the same way. Blocked or ruptured blood vessels my starve brain tissue of oxygen, so damaging a small area. In epilepsy, seizures begin with abnormal activity in a small area, which may spread to involve the whole brain.
Neurosurgeons have suggested that all these diseases are possible candidates for treatment with grafts of healthy brain tissue. There are no drugs available at present which can offer a cure.
The idea that grafts might work in human patients stems from experiments on laboratory animals. These show that grafts of nervous tissue cannot only survive in the brain of a laboratory animal, but also make connections with some of the host's cells .The grafts can even correct behavioural problems arising from certain types of brain damage.
This research has not only suggested a possible new treatment for human patients, but also increased our understanding of the brain's potential for growth and repair and shed some light on the way its functions are organised.
|
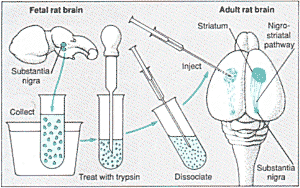
An adult rat with a damaged nigro-striatal pathway will turn circles.Cells from a fetal substantia nigra implanted into the striatum will once more set the rat on a straight course |
Elizabeth Dunn, an anatomist from Chicago University, demonstrated in 1917 that cells or pieces of nervous tissue could be successfully grafted from the brain of one new born rat to another. But it was not until the 1970s that researches in the US and Sweden improved on her rather hit-and-miss techniques by painstakingly establishing the conditions necessary for grated tissue to "take" in another brain. In particular, this means ensuring that the new tissue has access to a supply of blood, while avoiding its rejection by the immune system First, however, one has to choose appropriate tissue for grafting.
What to graft?
Cells on the grow
Traditional transplants involve surgeons taking the heart, a kidney or bone marrow, for example, from a living or dead donor, usually a mature adult. Things are very different with the brain. If the surgeon takes cells from a fully developed brain it disrupts their extensive connections. This is because the long fibres, or axons, which they use for communicating with other cells in the nervous system are damaged. Once the brain cells are disconnected, nothing can be done to keep them alive; they certainly will not survive grafting to another brain.
Unlike other tissues in the body, the nerve cells in the brain are more or less mature at birth. Only during the development of the foetus do the nerve cells have the capacity to increase their numbers by dividing to grow new axons over any distances and to make new connections.
Rejection |
The body's immune system provides an effective system of defence
against invasion foreign organisms, such as bacteria and viruses. The same
system normally ensures that any tissue not bearing the molecular label that
identifies it as "self" will automatically be attacked and destroyed (see
Inside Science Nos. 7 and 8). Patients receiving kidney or heart transplants
have to be treated with drug which suppress the immune system. The drugs
protect the transplant, but leave the patient vulnerable to infection. |
Most laboratory researches in neural transplantation, therefore, concentrate on taking tissue from the brains of rat foetuses and grafting it into adult rats. Cells in these grafts are not yet fully mature and have not established their connections. As a result, they are still flexible enough to cope with transplantation to another host.
Brain cells are not all alike; they come in a variety of shapes and sizes according to the regions they occupy and the tasks they have to perform. Each too, has a specific chemical identity. The cells of the nervous system communicate with one another by releasing a chemical, a neurotransmitter, from nerve endings at the tip of the axon. In its turn, the cell receiving the signal has specialised molecules called receptors on its surface. The receptors respond only to the appropriate neurotransmitter. The connection between cells take place at the synapse, where one cell released neurotransmitter across a tiny gap to another cell. Each cell has up to a thousand synapses.
Now that neuroscientists have mapped some of this immensely complicated chemical switching system, they can choose cells for grafting according to the neurotransmitter that the cells contain. Just as with a precision instrument, you can repair the brain more effectively if you have the right parts for the job.
In a typical experiment a graft consists of a few thousand cells, just a few cubic millimetres of tissue. This is inserted either as a solid piece or as a suspension of dissociated cells. Dissociation involves treating the cells with an enzyme, trypsin, to loosen to loosen their grip on one another and then shaking or stirring the tissue until it comes apart. The suspended cells are then drawn up into a syringe for injection. The size of the graft, and the method used to insert it, is one of the many factors that will determine whether it will "take" satisfactorily in its new host.
The New Home
Conditions for survival
For a graft to survive, its cells must quickly gain access to adequate amounts of oxygen and nutrients, normally by way of the hosts own blood supply. Early attempts at grafting often failed because experiments took a relatively large lump of cells and inserted it deep within the densely packed brain issue. The cells at the centre of the graft were starved of oxygen and soon died. New blood vessels from the host grew into the graft but too slowly to reach most of the tissue.
Smaller chunks of tissue stand a greater chance of success, while dissociating the cells greatly increases the graft's chances of survival. In addition injections of dissociated cells cause less damage to the host brain.
|
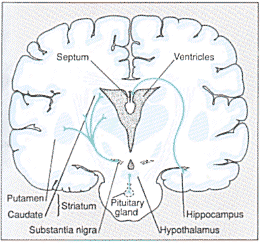
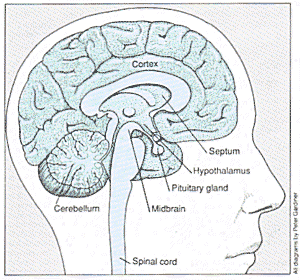
Three modulatory circuits or pathways, in a human brain. These pathways rely on neurotransmitters for communication. In experiments with rats and mice, grafts of cells tat produce the appropriate neurotransmitters have successfully corrected problems arising from damaged cells or faulty pathways. The cross-section of the human brain is idealised. In reality, the pituitary gland and striatum would to be visible; the pituitary gland is farther forward, and the striatum closer to the back of the brain. |
One way to improve the graft's chances of survival is to place it in one of the ventricles. These spaces within the brain are filled with cerebrospinal fluid, which appears to provide plenty of room for growth. The graft will attach itself to the wall of the ventricle and begin to grow fibres into nearby brain tissue. While this method causes relatively little damage to the host brain, the graft can reach only those areas adjacent to the ventricle.
Another option is to place the graft immediately below the cortex where there is a bed of densely interwoven blood capillaries. New blood cells quickly invade the graft, increasing the chances of the cells surviving. Again, the number of areas that can be reached by this technique is limited.
However, it is possible for neuro-scientists to create similar conditions at sites elsewhere in the brain. Removing a small amount of tissue from an implant site ahead of the grafting operation enables a new network of blood vessels to build up around the walls often artificial cavity. Researches have found that a graft implanted a week or two after the cavity is created is more likely to survive than one implanted immediately after. One theory suggests that the damage leads to build-up of special molecules called growth factors that stimulate and nourish the growth of new tissue.
Successful grafting also depends on the age of the donor cells. The ideal cells are those that have reached the stage when they are in the process of growing axons. This takes place at different points in gestation according to the area of the brain the cells come from.
In rats, the best age of tissue for grafting varied from about 11 days' gestation to just before birth 21 days after conception) depending on the type of cell. Cells taken from a foetus that is more than 1 days old, however, do not survive well if they are dissociated. Doctors do not agree about the ideal age at which they should remove human foetal tissue for treating patients. Experiments though, suggest that grafts taken from foetuses of between 9 and 2 weeks survive best.
Even under the best conditions, not all the cells in a graft will survive. In one experiment, research estimated that only between one in 100 and one in 1000 grafted cells survived and grew. The graft though, improved the rat's condition provided more than a hundred cells lived. In rats, therefore, one needs to inject at least 100 000 living cells to be sure of a good result. How large a graft needs to be to benefit a human patient is one of he many questions that need to be answered before doctors can consider he technique as a serious option in treatment.
What grafts do
Wiring or plumbing?
While the cells obviously have to survive if a graft is to play an active role in the host's brain, survival by itself is not enough. They have to establish some form of communication with other cells in the host brain. In the right circumstances, grafted cells will grow axons which penetrate short distances, making connections - they synapses - with cells in the surrounding issue. The graft may also stimulate cells in the host to grow new axons into it and make connections there.
Human cells for human brains: a new cure? |
||
The cells in a brain graft in laboratory animals survive. They
can also make connections with the host brain, and produce the neurotransmitter
for communication. On the basis of these experimental findings, doctors have
made a number of attempts to use grafts to treat human patients with brain
damage.
This has profound effects on people's ability to control their
muscles voluntarily. Patients are not paralysed, at least in the early stages
of the disease, but they may have difficulty beginning a movement. Their
limbs may also tremble uncontrollably or become rigid, and they may walk
stooped forward, with rapid shuffling steps. This disease tends to strike
in middle to old age. The symptoms worsen progressively over time, and there
is no cure at present. |
In the normal brain, communication between one region and another can take many different forms. Some circuits, or pathways, require transmission of precise information. One example is the visual pathway from the eye to the areas of the brain that make sense of what we see.
In other regions, one area may simply act to modulate the activity of another, rather like a volume control. In grafts that are to play a modulating role, the availability of the appropriate neurotransmitter would be more important than restoring the circuitry precisely. Normal movement for example, depends on connections between he subsantia nigra and he striatum an important motor control centre. The cells from the sustantia nigra send fibres to the striatum where they release the neurotransmitter dopamine. In a rat, an effect of damage to the nigro-striatal pathway on one side of the rain is to cause the animal to turn in circles.
A raft of cells from the susbstantia nigra of a foetus can reduce the rat's tendency to gyrate. Researches lace the graft, not at the side of the hosts substanita nigra, but directly into the striatum. There it makes some connections with the host cells, where it released dopamine. The graft enables the striatum once more to kept he rat on an even course. This is despite the abnormal position of the graft, and the fact that it lacks many of the connections of a normal substantia nigra.
This work has important implications for humans with Parkinson's disease. Cells in their substanita nigra have died, leading to a lack of dopamine in the striatum.
It is worth noting that this type of graft will form connections only if the host brain is missing its normal source of the transmitter. Receptor sites that are left empty can then be re-occupied by axons growing out from the graft. Animals gradually lose many of their brain cells as they age; in rates, at least, grafts can compensate for some of this loss.
Like elderly humans or patients with Alzheimer's disease, old rats have poor memories. The hippocampus plays an important role in storing new memories, receiving input from the septum via a pathway which uses acetylcholine as the neurotransmitter. The concentration of acetylcholine in the brain tends to decline with age.
Forgetful old rats given a graft of cells from a foetal septum can once more learn, and remember, their way around mazes. Again the precise circuitry is not important as along as the acetylcholine reaches the right target cells.
Some brain regions act as chemicals switches turning on the activity of another area. For example, the hypothalamus releases a hormone which in turn stimulates the nearby pituitary gland to produce more hormone. These act on the ovaries and testes, ensuring normal sexual development.
There is a strain of laboratory mouse which is genetically unable to produce the hypothalamic hormone, so neither males nor females mature sexually. A graft of foetal hypothalamic tissue from a normal mouse sets the hormone cascade into action; females with grafts have mated and given birth.
Recently, experiments with another type of graft have shown that it may be possible to rebuild circuits more precisely. Animals with damage to the striatum, which regulates movement, are overactive. They also have difficulty carrying out tasks such as retrieving food accurately with one or other paw. Grafts of striatal tissue can reduce these problems. To be efficient, these grafts have to be laced in the striatum itself, not as in the case of substania nigra grafts, in the areas with which it communicates.
If striatal grafts work, it must be because axons from the nigro-striatal and, possibly, other pathways are making contact with them and influence their activity. In addition, the graft's effectiveness should depend on its sending axons out into other areas and making connections there.
Researches who have studied the animal's brains under the microscope report that there are axons running to and from established striatal grafts. Their pattern suggests that they are indeed re-establishing the circuit that was broken when the striatum was damaged.
New directions
Future of brain grafts
The results of these and other experiments suggest that grafts could hold the promise of new treatments for humans. In particular, grafts might repair pathways that have a modulatory role. Neurosurgeons have yet conclusively to prove the effectiveness of brain grafts in humans however, and many practical questions remain to be resolved. In the meantime, other research is spawning new questions of its own. The techniques of genetic engineering mean that in theory, it is possible to tailor any cell in the body to fit different requirements. For example one could take a skin cell and later its genes so that it would make dopamine. Such cells will multiply happily in a laboratory dish and could then be used to as a graft to treat Parkinson's disease. This would have many advantages. Patients could donate their own skin cells, removing both the problem of rejection and the ethical difficulties of using tissue from foetuses.
Many find it hard to accept the idea of tinkering with the brain , which still holds many associations with the mind or personality. Such views may be out of date now that we are beginning to understand the workings of the brain well enough to consider repairing it. As for Dr Frankenstein's scheme of transplanting a whole brain to a new body - that remains in the realms of science fiction where it belongs.
© Reed Business Information 1999. Educational
copying of Inside Science is permitted under licence of Copyright Licensing
Agency Limited, 33 - 34 Alfred Place, London WC1 7DP
Back numbers of Inside Science are available
from New Scientist, P.O. Box 666, London ElS 1DW. Inquiries 0181
5030589.